Calina-IBd Study

Purpose of THe Study
The goal of this observational pilot study is to explore how inflammatory bowel disease (IBD) during pregnancy may influence the mother/birthing parent and infant gut inflammation and overall health. The study focuses on pregnant adults living in British Columbia, Canada.
Goals of calina-IBD study
The main questions this study seeks to answer are:
- How do gut microbiome profiles and dietary patterns during pregnancy differ between mothers/birthing parents with and without IBD?
- Are certain maternal/birthing parent gut bacteria or dietary habits linked to higher levels of gut inflammation in infants, as measured by fecal calprotectin (FCP)?
- How do infant gut microbiome and early diet (breastmilk) contribute to inflammation in the first year of life?
- How do sugars in breastmilk differ between mothers/birthing parents with and without IBD, and how might these sugars affect the infant’s gut bacteria and inflammation?
Researchers aim to identify diet, microbiome, and breastmilk-related factors that may contribute to your infant gut inflammation. These findings could help guide future dietary interventions to support healthier outcomes for mothers/birthing parent and their infant.
What is involved?
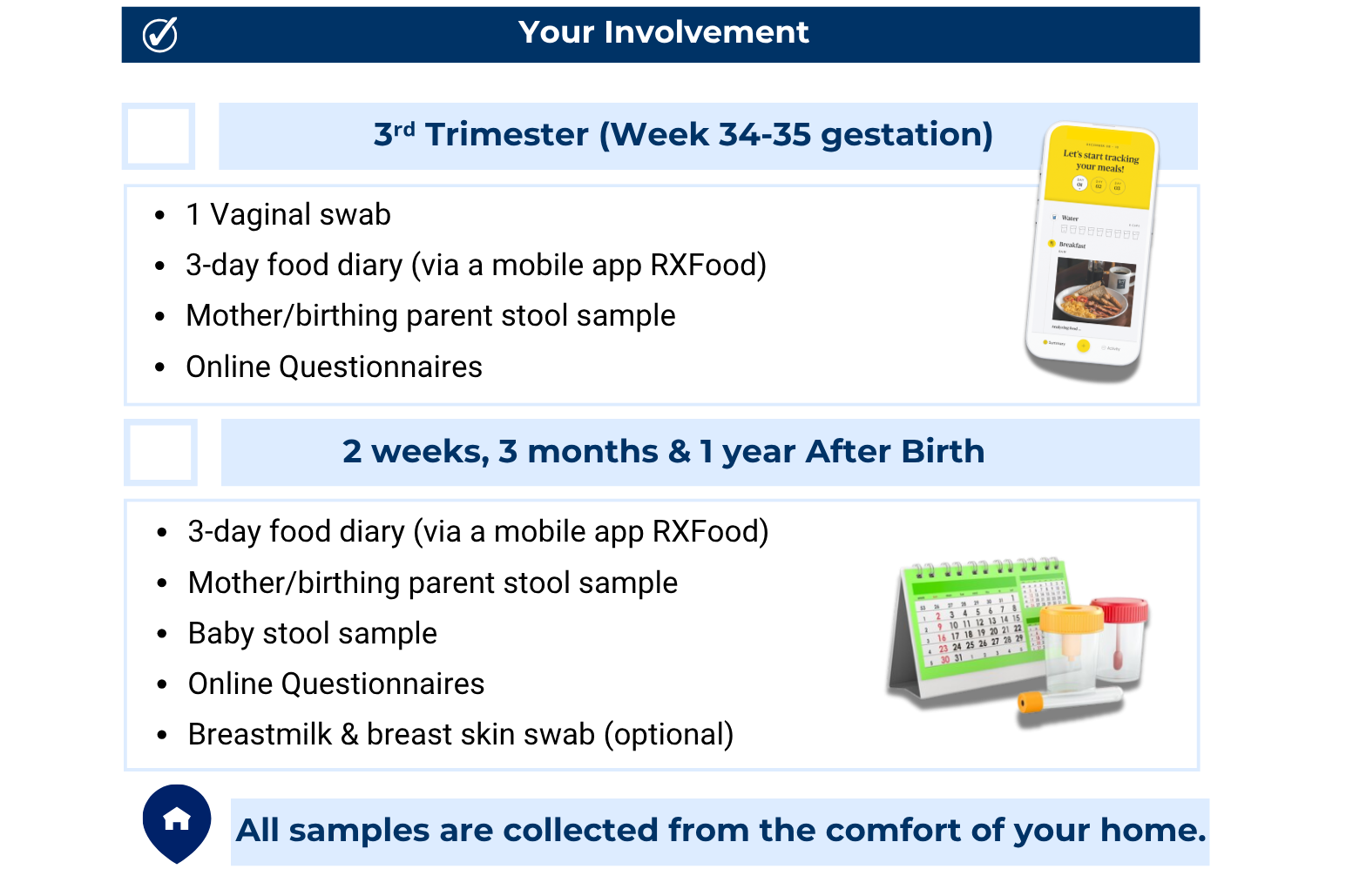
These samples and records will help researchers understand how maternal IBD, diet, microbiome, and breastmilk composition may influence infant gut inflammation and development. All materials will be provided and shipped by the research team, and participation can be completed from home.
Who can participate
Eligibility:
- Ages 19 years or older
- Confirmed pregnancy before 35 weeks of gestation
- For IBD group: Confirmed diagnosis of Crohn’s Disease, Ulcerative Colitis
- Interested in learning about bacteria in your gut
- For control: Do not have a diagnosis of IBD (serving as comparison group)
- Wanting to help advance research on maternal dietary interventions and early prevention for infants at high risk of inflammatory bowel disease
Exclusion:
- Travel outside of Canada and the United States in the month prior to first sample collection
- Inability to provide consent
- Previous gastrointestinal cancer or bowel surgery
- Renal disease
- HIV/AIDS or other serious infection
- Fetal chromosomal or structural abnormalities
- Other immune-mediated diseases (e.g., multiple sclerosis, rheumatoid arthritis, primary sclerosing cholangitis)
- Prebiotic, probiotic or postbiotic supplements in the month prior to first sample collection
- Gastroenteritis during or 1 month before the first sample collection
How the CALINA-IBD Study Could Benefit You
- Get free access to the RxFood App and receive personalized reports of your dietary intake at the end of the study (See example below)
- Learn about your gut microbiome and your baby’s microbiome
- Gain insight into your baby’s gut inflammation (measured by fecal calprotectin)
- Receive two $25 gift cards of your choice (e.g., Shoppers, Save-On-Foods, Amazon, Lululemon)
- Help us better understand the connections between diet, gut health, and early development
Contact us
If you are interested in participating in our study or would like to learn more, please contact us using the following methods below:
- Genelle Lunken, UBC, Principal Investigator
- Email: calina.ibd.ubc.ca
- Phone number: (+1) 604-441-4992
Learn more: Visit ClinicalTrials.gov For More Information
